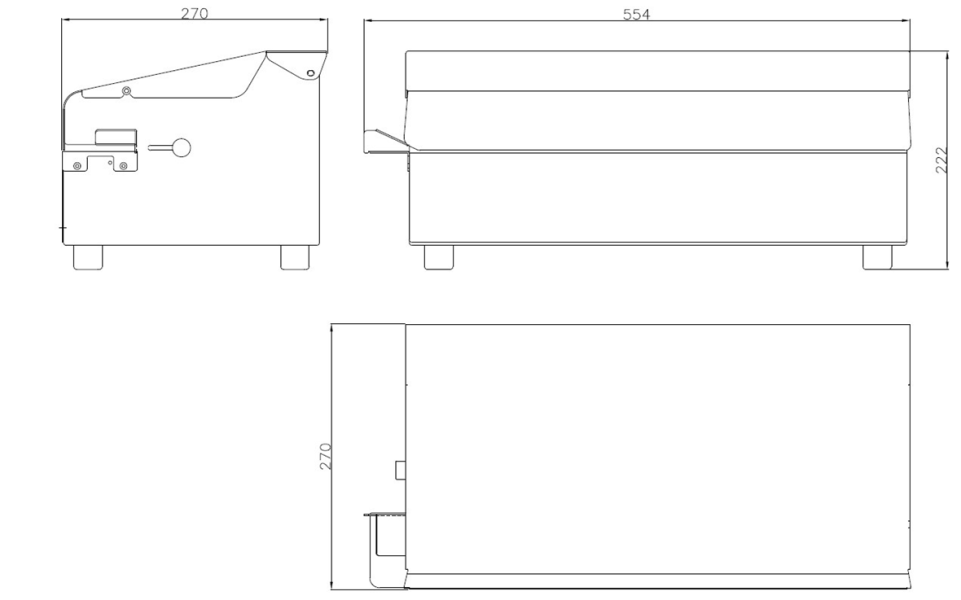
High-Tech performance and Validatable Sealing for Intensive Medical Device Packaging
ISO 11607.2 Validable Band Sealer with USB Port and SecurScan Data Upload Software.
Engineered for hospitals, CSSDs, and industrial facilities that demand precision, compliance, and full traceability in sterile packaging.
Key Technical Features
✅ Sealing width 15 mm in accordance with DIN 58953.7 standards.
✅ Heat-sealable materials: EN 868-5 pouches and rolls, TYVEK pouches, EN 868-4 paper/paper pouches, gusseted formats, PP laminated aluminum.
✅ Sealing speed up to 10 m/min.
✅ Temperature control 10–200°C with ±1% tolerance.
Compliance with World Health Organization Guidelines and World Federation for Hospital Sterilization Sciences (WFHSS)
Key Benefits
Precision & Process Validation
Next-generation hardware and software for real-time, precise control of temperature, pressure, and sealing speed.
Fully validable according to ISO 11607.2 and EN-ISO TS 16775, guaranteeing consistent, repeatable, and traceable packaging processes.
With a user-friendly, intelligent interface that accelerates training and ensures error-free operation.
Safety
Sealing height mm 15 for correct peelability and maximum operational safety for sterile barrier systems.
Automatic process monitoring with alerts for out-of-range parameters.
Advanced Operator Interface
4-line LCD display (20 characters per line) with flex strip alphanumeric membrane keyboard for intuitive operation.
Clear visual indicators for status, warnings, and parameter adjustments.
Gandus minirò H-net user-friendly, intelligent interface ensures efficient operation and reduces the risk of errors.
Efficient connetivity and integration
RS-232 interface for PC connection, label printer, and barcode scanner.
USB port for fast, safe, and cost-free data download, traceability, and software updates without requiring proprietary modules or additional infrastructure.
Fully compatible with Securscan Data Upload Software for barcode generation, parameter logging, and easy integration into hospital IT workflows.
Codable print
Built-in single-line printer.
All print data can be encoded on board the machine and immediately recalled from memory.
Full and efficient traceability
Full, simple, safe traceability of each sealed pouch via USB export to pen drives.
No costs for additional hardware as traceability files are downloaded to storage pen drives.
The USB port also allows for simple, fast and safe software updates for the machine.
Securscan Data Upload Software
Gandus Saldatrici data loading system, extremely safe and easy to use, able to read the sealing data for each sealed bag and to automatically define the machine functions and parameters.
The Securscan Data Upload Software automatically reads sealing parameters, generates barcodes with all sealing parameters for each pouch ID, and transfers data to any PC for complete process documentation.
The system is extremely simple to install and works on any PC.
Sanitization at the highest levels
AISI 304 stainless steel cover for easy cleaning and compliance with hospital-grade sanitization protocols.
Energy Efficiency
Optimized for optimized energy consumption without compromising performance or sealing quality.
Materials
EN 868-5 pouches and rolls, TYVEK envelopes, paper/paper pouches EN 868- 4, with gussets, Laminated aluminium, PP.
Accessories
PS 654 worktop
RL 80 roller conveyor
PR H40 roll holder complete with cutting device
PR H50 roll holder complete with cutting device
PRN 2000 label printer
SecurScan Data Upload Software
Regulatory Compliance
UNI EN ISO 11607-2
UNI EN 868-5
EN ISO TS 16775
DIN-58953 7
Directives 2006/42/EC-2014/30/UE-2014/35/UE
UNI/TR 11408
Sterilization Guidelines-MEMO 5
Manufactured in an UNI EN ISO 9001:2015 Quality System Certified Company
Testing and Validation Services
Gandus Saldatrici offers a range of Testing and Validation Services to ensure safe and reliable daily monitoring of medical sealing machines for sterilization pouches in accordance with ISO 11607.1.2 and ASTM F1929 Standards. learn more