Utmost Precision. Total Monitoring and Control.
Zero compromises in the ultimate space-saving design.
Pura represents the ultimate synergy of cutting-edge technology, regulatory compliance, and essential design. Gandus Pura heat sealers are engineered for unmatched operational reliability, consistent seal quality, and full control over process parameters.
Developed to meet the highest international standards for medical packaging validation, Pura guarantees reliable, repeatable, and fully monitored and traceable sealing performance for sterile barrier systems.
Compact design, clean lines, and robust construction combine with advanced hardware and software architecture to provide:
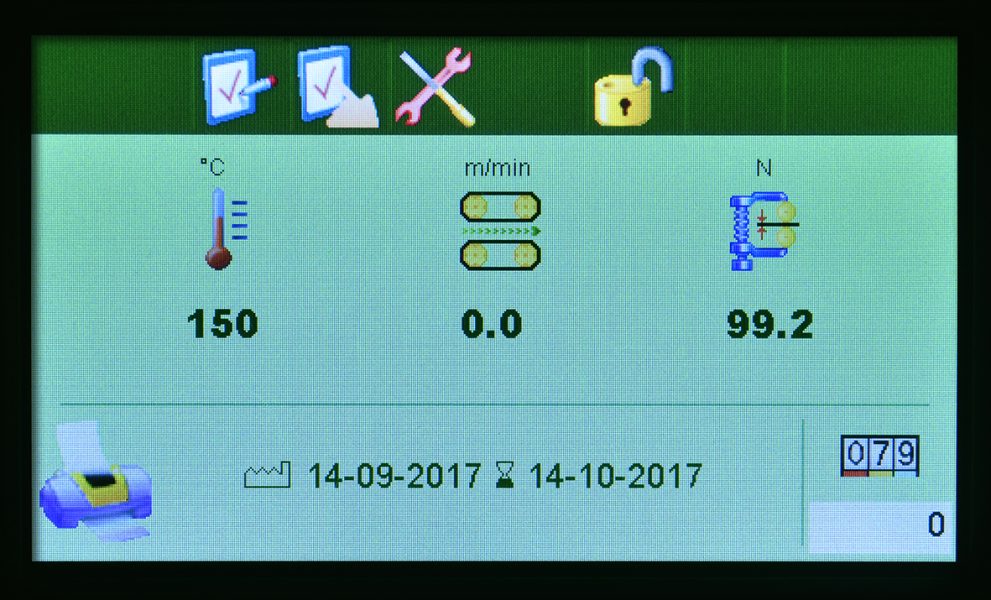
✔ Precise control of temperature, pressure, speed
✔ Uniform sealing in every cycle
✔ Cost optimization
✔ Easy integration into existing production lines and external systems
✔ Integrated printing device
✔ Extensive, flexible, and modern connectivity
Designed for hospitals, CSSDs, medical device, pharmaceutical, and LifeScience environments, Pura ensures accurate temperature control, complete process traceability, and intuitive operation. Its clean lines, premium materials, and compact footprint make it the ideal solution where hygiene, efficiency, and space optimization are essential.
Certified performance for high-criticality environments
• Process traceability
• Certified repeatability
• Compliance with industry regulations
• Operational stability even under intensive production
Every detail is engineered to deliver long-term reliability and reduce total cost of ownership.
More control. More efficiency. More value.
• Greater configuration flexibility
• Intuitive operator interface
• Simplified maintenance
• Direct and timely technical support
Compliant with ISO 11607.2 and major international sterilization guidelines, Pura embodies validated performance through purity of design, engineering precision, full monitoring and control. To get to uncompromising safety.
Key Technical Features
✅ ISO 11607.2 Validatable Medical rotary heat sealer.
✅ Management from 4.3” color touch screen.
✅ Essential and clean lines.
✅ AISI 304 stainless steel.
✅ Heat Sealable Materials: Heat Sealable Sterile Barrier Systems, Tyvek®, Next Generation Packaging Materials.
✅ Rotary sealing, mm 15 height .
✅ Sealing speed 10 m/min.
✅ Maximum sealing temperature 200°C, tolerance +/- 1%.
✅ Compliance with World Health Organization guidelines, World Federation for Hospital Sterilization Sciences (WFHSS).
Why Pura stands out.
Reliable and Compliant Packaging Every Time
Next-generation hardware and software deliver precise temperature, pressure, speed monitoring and control, fully compliant with ISO 11607.2 and international guidelines, ensuring validated performance in every seal.
Secure Seals, Safe Handling
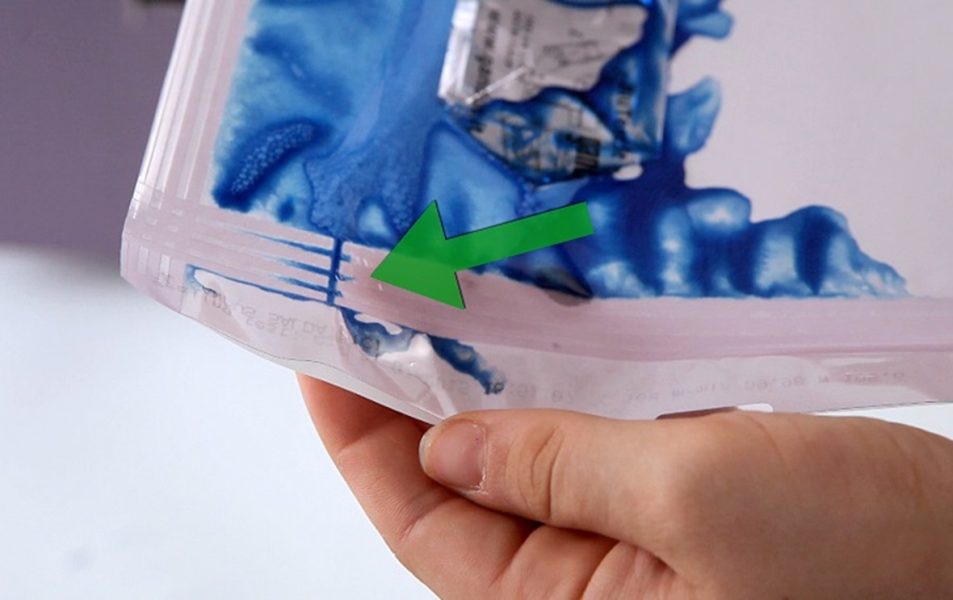
mm 15 multi-line sealing ensures perfect seals and easy, safe pouch opening for consistent, reliable sterilization results.
Quick Maintenance, Maximum Efficiency
Built-in entry guides and dual access (top and front) allow fast, ergonomic maintenance, reducing downtime and effort.
Seamless Connectivity for Smarter Workflow
USB, RS232 and included Ethernet interfaces enable smooth integration with external systems, streamlining data exchange and operational efficiency.
Full Traceability at Your Fingertips
All process data are automatically recorded to standard USB media, ensuring complete traceability and effortless compliance reporting.
Effortless Operation for Maximum Safety
Large 4.3” colour touch screen and dedicated software provide an intuitive interface that simplifies tasks, reduces errors, and ensures safe, efficient operation.
Fast Data Management with SecurScan Data Upload Software
SecurScan Data Upload Software allows quick download of all machine data via scanner, simplifying monitoring and recordkeeping.
SmartPrint. Accurate Printing, Optimized for Every Pouch
SmartPrint needle printer automatically adjusts font size to pouch dimensions, with alarm in case of too small pouch, prints text and barcodes efficiently, and provides quick cartridge changes.
Compact Design, Maximum Productivity
Pura requires only 0.15 m², delivering full sealing capability in a minimal footprint for space-optimized work environments.
Hygienic Design for Safe Environments
Stainless steel, anodized aluminum, and clean, ergonomic lines allow easy cleaning and disinfection to the highest standards.
Energy-Efficient Operation
Engineered for process optimization with minimal energy consumption, combining sustainability with high performance.
NEW! Flexible Firmware for Seamless Updates
– copy operator, batch and catalog libraries stored on a USB stick
– perform a quick search for a catalog string by typing the initial letters of the string.
– the new firmware can be implemented on all machines produced so far via USB stick.
Materials
Ideal for all heat-sealable sterile barrier systems, Tyvek®, and next-generation packaging materials, ensuring versatile and reliable sealing.
Versions
Stainless steel and anodized aluminum version
Stainless steel version
No Print version (Pura NP)
Accessories on request
Work plan
Roller tray
Roll holder
Label printer
Barcode scanner
Ethernet module
Regulatory Compliance
UNI EN ISO 11607-2
UNI EN 868-5
EN ISO TS 16775
DIN-58953 7
Directives 2006/42/EC-2014/30/UE-2014/35/UE
UNI/TR 11408
Sterilization Guidelines-MEMO 5
EU GMP Annex 1: manufacture of Sterile Medicinal Products
Manufactured in an UNI EN ISO 9001:2015 Quality System Certified Company
Testing and Validation Services
Gandus Saldatrici offers a range of Testing and Validation Services to ensure safe and reliable daily monitoring of medical sealing machines for sterilization pouches in accordance with ISO 11607.1.2 and ASTM F1929 Standards. learn more
Pura. Validatable Perfection through Pureness.
Product design and engineering developed with the scientific consultancy of the Design Department of the Politecnico University of Milan.