ISO 11607.2 compliant Pouch Production System for Medical Applications
Engineered to Ensure Compliance, Traceability, and Full Process Control
High-precision highly configurable automatic Seal-Cut production system engineered to fully customize each individual pouch with V-seal (Chevron seal), starting from standard roll.
The system enables configurable pouch dimensions (length and width) and controlled batch production while maintaining strict process reproducibility.
The machine architecture allows integration within regulated manufacturing environments, facilitating compliance with international standards for sterile barrier systems and validated sealing processes.
The control of critical process parameters, alarm management, and interface with traceability systems help ensure process repeatability, documentation, and batch integrity, all of which are essential for regulatory compliance in the medical device sector.
Key Technical Features
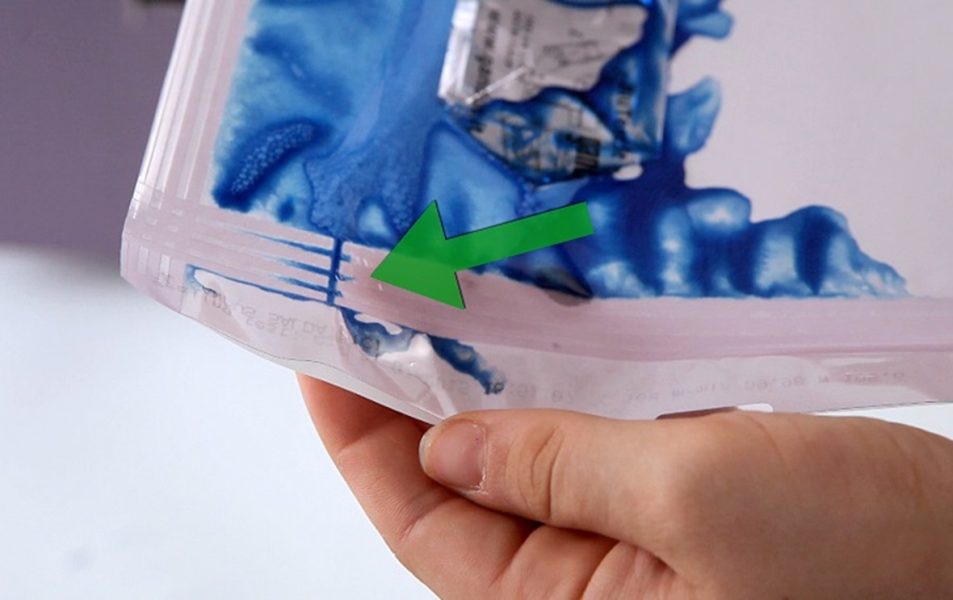
✅ Multi-line V-shaped chevron seal with 150° angle.
✅ Sealing height 11,5 mm.
✅ Configurable pouch dimensions (length and width) and controlled batch production
✅ Control of critical process parameters
✅ Interface with traceability systems
✅ Alarm management
Key Benefits
Highly configurable production system designed to customize each individual pouch in terms of length, width, and batch size, while ensuring full process repeatability.
Controlled Efficiency and Footprint Optimization
Compact mechanical layout optimized for space-constrained cleanroom environments.
Equipped with an automatic roll centering system controlled via HMI to minimize operator variability and setup time.
Production capacity up to 20 pouches per minute, with stable, repeatable and validation-ready performance.
Advanced Sealing Configuration
The proprietary sealing design enables the production of pouches featuring single-side V-sealing starting from standard roll materials.
This configuration maintains material barrier integrity and ensures consistent seal performance, supporting sterile barrier system requirements.
Sealing Process Control
The sealing unit is managed by an automatic control system ensuring continuous monitoring and regulation of critical process parameters, including:
sealing temperature
applied sealing pressure
sealing dwell time / cycle speed
Each parameter operates within validated tolerance windows. Any deviation outside predefined acceptance criteria triggers immediate cycle interruption and a visual/audible alarm.
This ensures non-conforming product prevention and batch integrity protection.
Interface with traceability systems.
High productivity, the machine can produce up to 20 bags per minute.
Accessories on request
Worktop complete with pouch tray
Regulatory Compliance
The system is designed to operate in accordance with the regulatory requirements applicable to medical packaging, supporting validated, controlled, and fully traceable processes.
UNI EN ISO 11607-2
UNI EN 868-5: 2019
EN ISO TS 16775
DIN-58953 7
Directives 2006/42/EC-2014/30/UE-2014/35/UE
UNI/TR 11408:2011
Sterilization Guidelines-MEMO 5
Manufactured in an UNI EN ISO 9001:2015 Quality System Certified Company
Testing and Validation Services
Gandus Saldatrici offers a range of Testing and Validation Services to ensure safe and reliable daily monitoring of medical sealing machines for sterilization pouches in accordance with ISO 11607.1.2 and ASTM F1929 Standards. learn more