Smart Impulse Sealing with Advanced Parameter Control Technology
Impulse heat sealers providing complete control over all sealing parameters, ensuring a Validatable Sealing Process in full compliance with ISO 11607-2, ISO TS 16775, EN 868-2, and DIN 58953-7 standards.
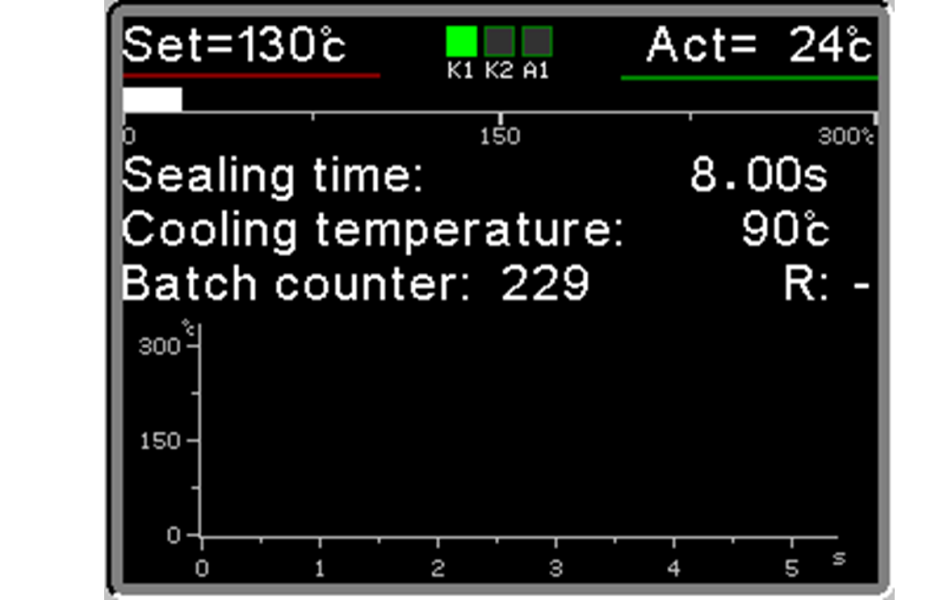
Today equipped as standard with the new Ropex RESISTRON® RES 5450 Controller, delivering enhanced performance and precision.
The machine features an AISI 304 stainless steel cover for maximum hygiene and ease of cleaning.
Key Technical Features
✅ Sealing type: Impulse, flat
✅ Sealing height: mm 8
✅ Max sealing lengths mm 460/610/710
✅ Heat-sealable materials: Sealable sterile barrier systems, Tyvek®, and new-generation materials
✅ Operation manually or via optional electric foot switch
Key Benefits
Packaging Process Validation
Medical H R25 are designed for high-precision control of all sealing parameters—Temperature, Pressure, Time, which are continuously monitored 50 times per second by the RESISTRON® RES 5450 Controller. This guarantees absolute consistency, reproducibility, and compliance with ISO 11607-2 and international guidelines EN-ISO TS 16775.
If any parameter exceeds its preset tolerance, the machine automatically stops and displays an alarm. This ensures that every seal is perfectly reliable and verifiable.
Intuitive High-Precision Operation and Control
The machine’s user LCD interface is simple, immediate, and ergonomic, allowing intuitive operation without extensive training. Operators can easily set parameters, monitor the process, and adjust in real time, combining usability and operational speed with high-precision control. For Maximum Efficiency.
Safety and Reliability
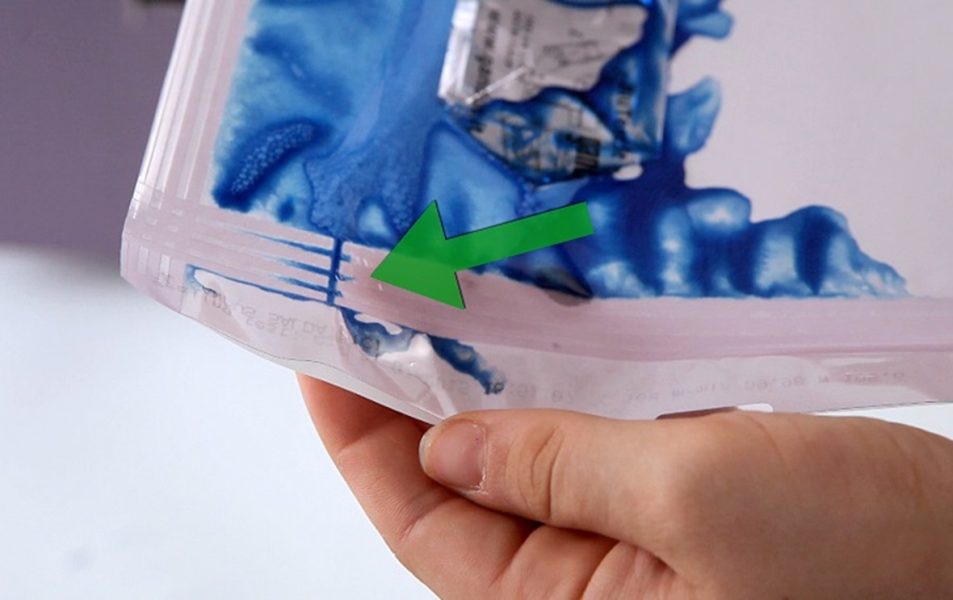
A flat 8 mm seal ensures optimal sealing performance and effective peelability, combining reliability with safety for sterile packaging.
Communication & Traceability
✓ USB-C interface for seamless connection with dedicated traceability software
✓ dedicated traceability Remote Management Software enables complete monitoring, configuration, and data logging via a personal computer. All sealing data are recorded, stored, and easily retrievable, supporting measurable, verifiable, and auditable processes.
USB-C connection and remote management software allow full data logging, ensuring measurable and verifiable sealing processes.
Hygienic Design for Maximum Safety
Stainless steel cover supports rapid cleaning and disinfection, ideal for sterile and clinical environments.
Key Benefits at a Glance
✅ High-precision sealing
✅ Intuitive, ergonomic interface
✅ Rapid and efficient operation
✅ Full traceability & audit-ready data logging
✅ Safe, reliable, and hygienic seals
Materials
Designed for sealing all sealable sterile barrier systems, Tyvek®, all new generation packaging materials.
Available Versions
Sealing lengths mm 460 – mm 610 – mm 710 – mm 810
Standard Version and GT Version, with built-in roll holder and cutter. Cutting length mm 370/500/610/610.
Available Accessories
Work plan
PR H40 roll holder complete with cutting device
PR H50 roll holder complete with cutting device
Thermometer and probe kit for temperature calibration
Regulatory Compliance
UNI EN ISO 11607-2
UNI EN 868-5
DIN-58953 7
2006/42/EC-2014/30/UE-2014/35/UE
UNI/TR 11408
EU GMP Annex 1: Manufacture of Sterile Medicinal Products
Manufactured in an UNI EN ISO 9001:2015 Quality System Certified Company
Testing and Validation Services
Gandus Saldatrici offers a range of Testing and Validation Services to ensure safe and reliable daily monitoring of medical sealing machines for sterilization pouches in accordance with ISO 11607.1.2 and ASTM F1929 Standards. learn more