ISO 11607-2 Validatable Rotary Heat Sealer. Precision. Compliance. Confidence.
Engineered for environments where sterility is non-negotiable, this advanced rotary heat sealer delivers validated performance, complete traceability, and uncompromising seal integrity — fully compliant with ISO 11607-2.
Designed for medical device manufacturers and sterile processing departments that demand excellence without compromise.
Key Technical Features
✅ 9 mm knurled sealing band (flat version available as option)
✅ Controlled sealing speed: 6 m/min
✅ Optional speeds: 4 m/min – 8 m/min
✅ Temperature range: 10°C – 220°C
Wide material compatibility
EN 868-5 pouches and reels
Tyvek® pouches
EN 868-4 paper/paper pouches
Gusseted pouches
Laminated aluminum pouches
PP pouches
Header bags
Polyethylene
Why It Stands Out
Absolute Process Control
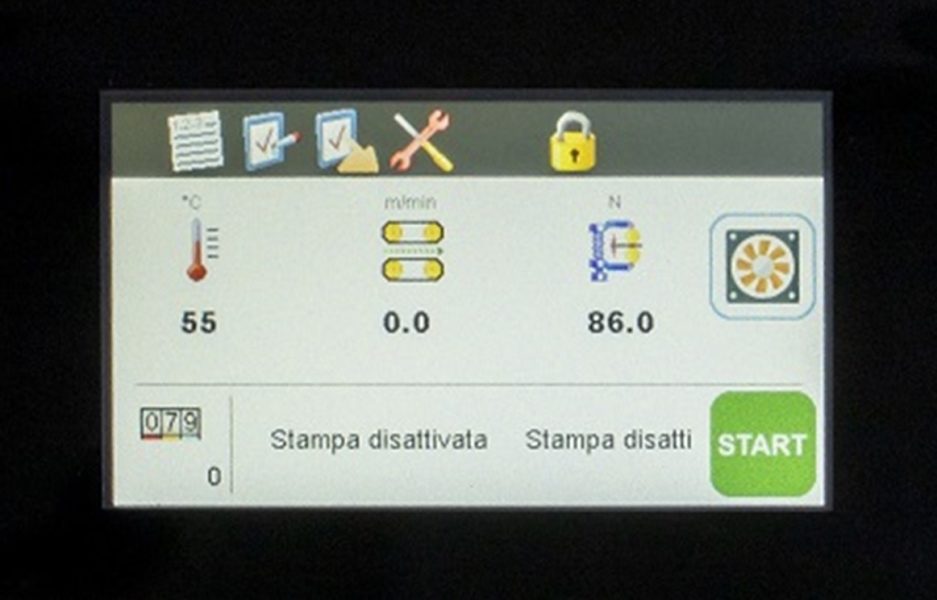
Real-time monitoring and precise control of critical sealing parameters (temperature, speed, pressure) ensure consistent, repeatable seal quality over time and support full Validation protocols and international compliance requirements as ISO 11607-2 and EN ISO TS 16775.
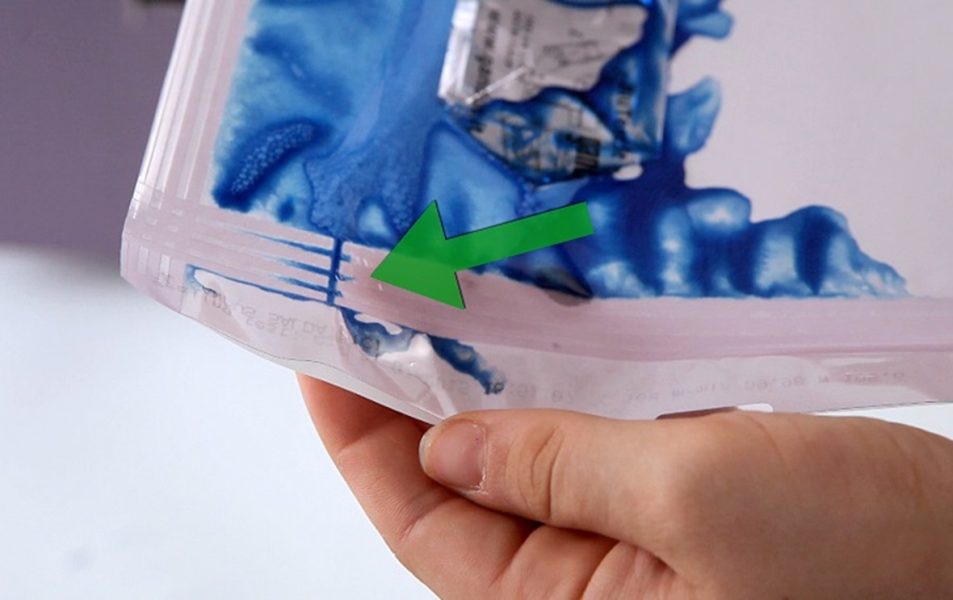
Reliable Seal Integrity & Safe Peelability
mm 9 knurled sealing (flat option) to ensure a uniform mechanical strength, optimal barrier performance, controlled and safe peel opening.
Advanced Operator Interface
4.3” high resolution color touch screen provides a clean, usr-focused interface for recipe management, parameter configuration, real-time system monitoring.
Full Traceability Management
Integrated data recording system enabling sensitive data tracking of each sealed pouch with USB export capability.
Designed to support audit readiness and documentation requirements.
Flexible Connectivity for efficient workflow integration
USB port for data upload/download and firmware updates.
RS 232 interface for barcode reader, label printer, and remote PC connection.
Efficient printing
Direct integration with label printer for fast, accurate, and compliant identification of sterile barrier systems.
Hygienic by Design
AISI 304 stainless steel housing ensures superior cleanability and suitability for controlled environments.
Energy-Optimized Performance
Optimized design to reduce power consumption while maintaining high operational performance.
Compatible Materials
EN 868-5 bags/rolls, TyvekR bags, EN 868-4 paper/paper bags, gusset bags, laminated aluminium bags, PP bags, header bags, polyethylene.
Available Accessories
Motorized conveyor belt
PS 654 workt table
RL 80 sliding roller conveyor
Barcode Reader
PRN 2000 label printer
Regulatory Compliance
UNI EN ISO 11607.2
UNI EN 868-5
EN ISO TS 16775
DIN-58953 7
Direttive 2006/42/EC-2014/30/UE-2014/35/UE
UNI/TR 11408
Sterilization Guidelines-MEMO 5
Manufactured in an UNI EN ISO 9001:2015 Quality System Certified Company
Testing and Validation Services
Gandus Saldatrici offers a range of Testing and Validation Services to ensure safe and reliable daily monitoring of medical sealing machines for sterilization pouches in accordance with ISO 11607.1.2 and ASTM F1929 Standards. learn more