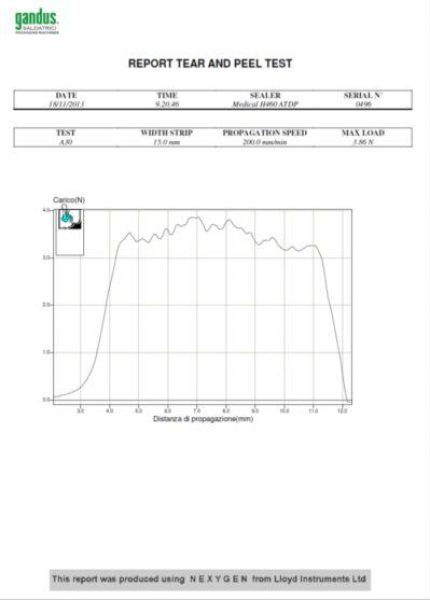
Heat-Sealed Joint Integrity Testing in accordance with EN 868-5 and Performance Qualification (PQ) procedures
Compliance with EN 868-5 requires the execution of specific tests on heat-sealed seams to verify the mechanical integrity and performance of medical packaging systems.
The EN 868-5 standard defines:
-
The performance requirements for medical paper/plastic pouches and reels
-
The control methods applicable to finished products
-
The test procedures to be performed on heat-sealed seams
Among these, particular relevance is given to the verification of seal strength and integrity to ensure that packaging systems maintain sterility throughout handling, transport, and storage.
Using calibrated measuring instruments and controlled test methods, Gandus Saldatrici provides professional Peel Strength Test services on heat-sealed joints, ensuring:
-
Objective measurement of seal strength
-
Verification of compliance with EN 868-5 requirements
-
Support for documented evidence within PQ validation protocols
-
Full traceability of test results
These activities contribute to confirming that the sealing process, under validated operating parameters, consistently produces compliant and reliable seals.
Our technical specialists are available to provide expert assistance tailored to your application requirements.